Dive Brief:
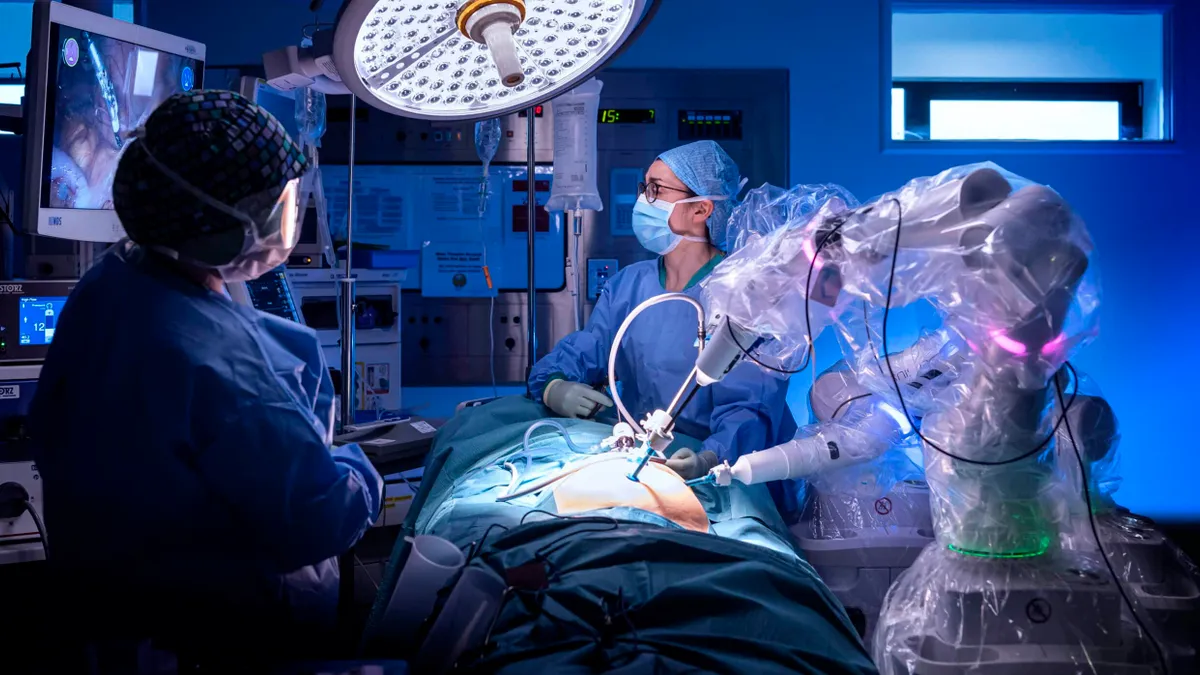
- CMR Surgical said Wednesday it closed a financing round worth more than $200 million to support commercializing the robot maker’s Versius soft tissue system.
- The Cambridge, U.K.-based company now has its sights on the U.S. market, after winning Food and Drug Administration de novo authorization for the robot in October with an initial indication for gallbladder removal surgery.
- CMR plans to use the new funds to expand its worldwide rollout of Versius to hospitals, with a “major focus” in the U.S., and to advance innovation and product development in surgical robotics.
Dive Insight:
CMR, one of multiple companies competing for a portion of the growing market in robotically assisted surgery, claims that Versius is the second most utilized soft tissue robot worldwide, after industry leader Intuitive Surgical’s da Vinci system.
Mark Slack, CMR co-founder and chief medical officer, told MedTech Dive last year that new competition will be healthy for the market. The company built its base in global markets before preparing to compete with Intuitive on its home turf in the U.S., Slack said.
CEO Massimiliano Colella, in a statement Wednesday, said CMR is focused on further penetration of existing markets in addition to the U.S. launch.
“We are now at a pivotal stage, poised to capitalise on significant opportunities for market expansion,” Colella said.
Versius has been used to complete more than 30,000 surgical cases globally in more than 30 countries, across specialities including colorectal, general, gynecology, thoracic and urology surgeries. The company is also studying use of the robot in pediatric surgery and other applications.
The funding round combines equity and debt capital and is backed by new investor Trinity Capital and existing CMR investors, the company said.