Cardiac: Page 3
-
J&J coordinating with FDA on US pause of Varipulse due to safety risks
Johnson & Johnson executives, on a Wednesday earnings call, did not provide a timeline for when U.S. Varipulse cases will restart after halting procedures in early January.
By Ricky Zipp • Jan. 22, 2025 -
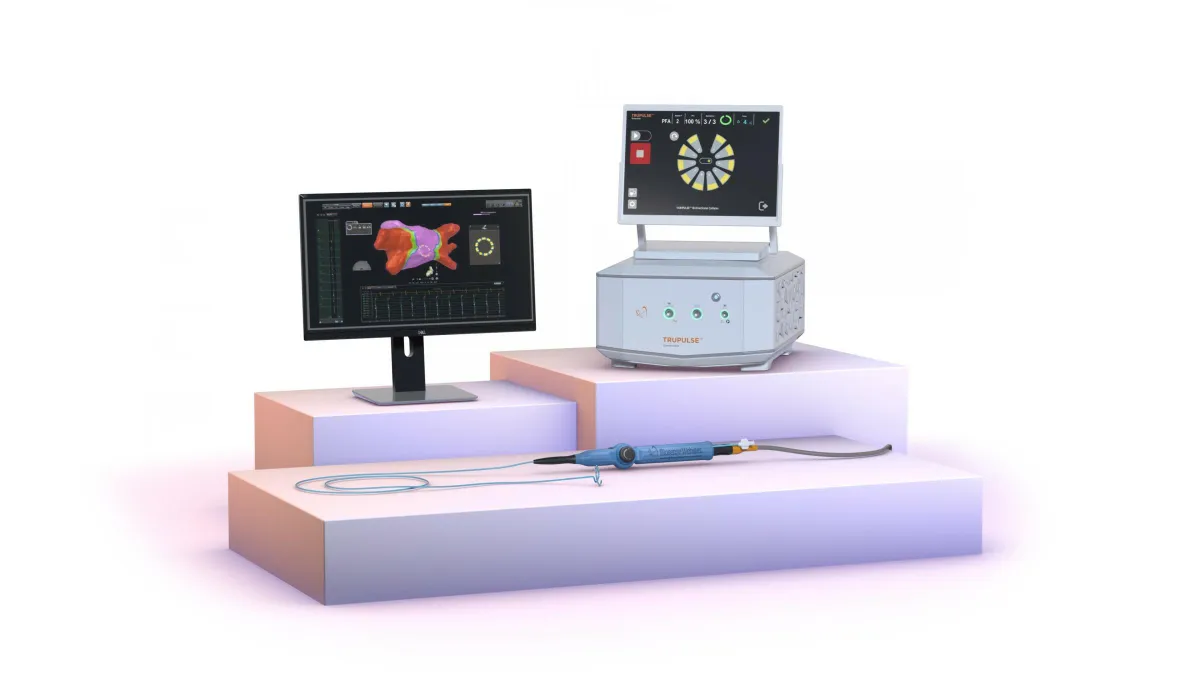
Boston Scientific study meets goals for PFA in persistent AFib
The results will support the company’s application for expanded labeling of its popular Farapulse pulsed field ablation system in a larger group of patients with atrial fibrillation.
By Susan Kelly • Jan. 21, 2025 -
 Explore the Trendline➔
Explore the Trendline➔
 Sarah Silbiger via Getty Images
Sarah Silbiger via Getty Images Trendline
TrendlineMedical device safety in spotlight after high profile recalls
From Philips’ massive recall of respiratory devices to ongoing health risks with breast implants, medical devices tied to patient harm have put a focus on product safety.
By MedTech Dive staff -
JPM25 recap: Robotics, PFA and OTC glucose sensors in the spotlight
CEOs from Medtronic, Intuitive Surgical, Boston Scientific and Dexcom were on hand this week in San Francisco to discuss the year ahead at the J.P. Morgan Healthcare Conference.
By Susan Kelly • Jan. 17, 2025 -
Boston Scientific ups PFA forecast, targets market leadership
At the J.P. Morgan Healthcare Conference, Boston Scientific forecast that pulsed field ablation will be used in more than 60% of atrial fibrillation procedures globally by 2026.
By Nick Paul Taylor • Jan. 15, 2025 -
Philips recall of heart monitor software tied to 109 injuries, 2 deaths
Some electrocardiogram events, such as atrial fibrillation or abnormally rapid heartbeats, over two years were not reviewed by healthcare professionals due to a data routing issue.
By Ricky Zipp • Jan. 14, 2025 -
CEO Geoff Martha tells JPM crowd Medtronic is ‘stacking growth drivers’
New technologies for atrial fibrillation, hypertension, diabetes and neurological conditions are poised for fast growth, Martha said Monday at the J.P. Morgan Healthcare Conference.
By Susan Kelly • Jan. 14, 2025 -
CMS starts Medicare coverage analysis for Medtronic, Recor renal denervation devices
J.P. Morgan analysts said the CMS’ review “validates the technology and could potentially move the needle on growth for Medtronic.”
By Nick Paul Taylor • Jan. 14, 2025 -
Eko Health names Justin Spencer as CFO
Spencer’s arrival at Eko comes months after the digital health company appointed his former colleague Brent Lang to its board of directors.
By Nick Paul Taylor • Jan. 10, 2025 -

 Retrieved from Fire1 on January 10, 2025
Retrieved from Fire1 on January 10, 2025
Fire1 raises $120M to trial heart failure management system
Medtronic and Novo Holdings were among the investors in the funding round.
By Nick Paul Taylor • Jan. 10, 2025 -
Deep Dive // 2025 outlook
5 medtech trends to watch in 2025
After a busy 2024, experts called out competition in soft tissue robotics, uncertainty from a Trump White House and continued success for pulsed field ablation as trends to watch this year.
By Ricky Zipp • Jan. 9, 2025 -
J&J pauses Varipulse PFA cases in US
The temporary halt to investigate reported neurovascular events comes two months after Johnson & Johnson won FDA approval for the pulsed field ablation system.
By Susan Kelly • Jan. 8, 2025 -
Boston Scientific to buy remaining Bolt Medical stake for up to $664M
The deal for Bolt, developer of an intravascular lithotripsy system to treat coronary and peripheral artery disease, continues Boston Scientific’s brisk run of acquisitions.
By Susan Kelly • Jan. 8, 2025 -
Top 10 medtech deals of 2024
Multibillion-dollar buyouts returned last year, led by Johnson & Johnson’s $13.1 billion acquisition of Shockwave Medical.
By Nick Paul Taylor • Jan. 2, 2025 -
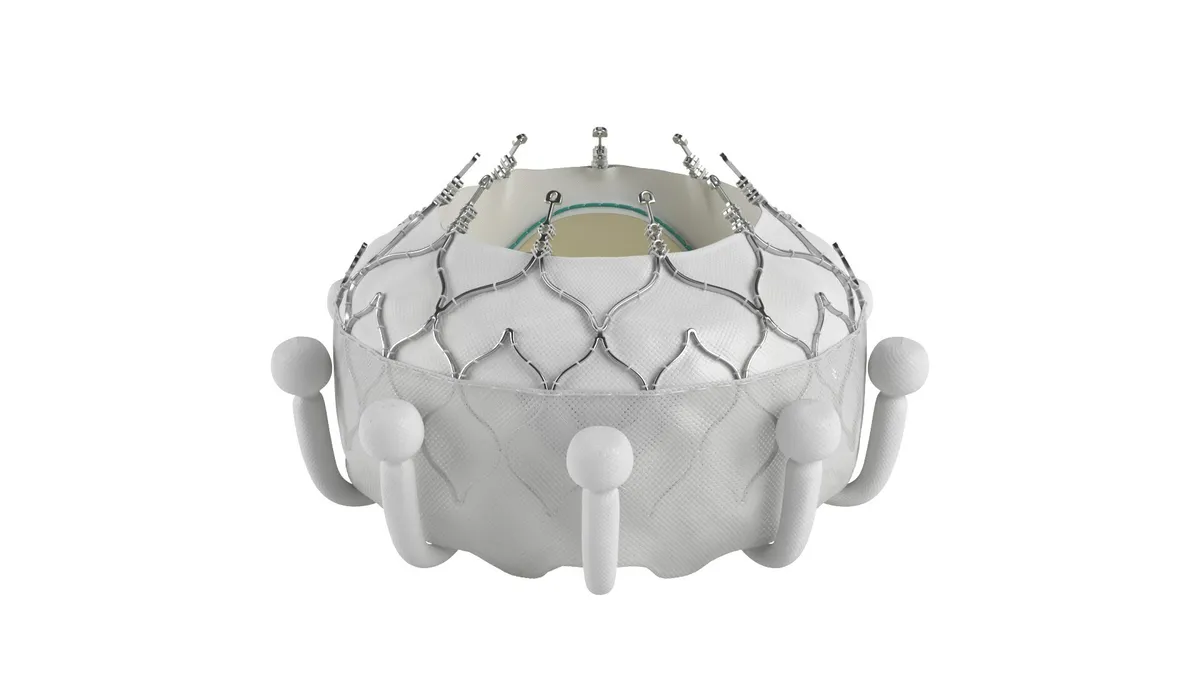
CMS proposes Medicare coverage for transcatheter tricuspid replacement
Edwards submitted a request for a national coverage determination in February, after receiving FDA approval for its Evoque device earlier that month.
By Elise Reuter • Updated Dec. 23, 2024 -
Robots, AI and PFA: The top medtech Q&As of 2024
From challenging market leaders to the growing influence of artificial intelligence, medical device executives had a lot to discuss this year. Check out MedTech Dive’s top Q&As of 2024.
By Ricky Zipp • Dec. 20, 2024 -
Heart valve developer Anteris launches IPO
IPOs in medtech have been sparse, but some analysts said improving market conditions could support a revival of offerings in 2025.
By Susan Kelly • Dec. 20, 2024 -
Boston Scientific updates cryoablation catheter instructions after 4 death reports
The update followed a higher than expected number of reports of esophageal injury after catheter ablation procedures for atrial fibrillation.
By Nick Paul Taylor • Dec. 19, 2024 -

 Retrieved from Merit Medical on December 17, 2024
Retrieved from Merit Medical on December 17, 2024
Merit Medical president to resign after unspecified conduct allegations
Joseph Wright’s resignation is effective in January, and CEO and founder Fred Lampropoulos has been reappointed president.
By Susan Kelly • Dec. 18, 2024 -
PFA to surpass radiofrequency ablation in 2025: Citi survey
Pulsed field ablation could soon be used in the majority of electrophysiology procedures to treat atrial fibrillation as Medtronic, Boston Scientific and Johnson & Johnson battle for market share.
By Susan Kelly • Dec. 16, 2024 -
Capstan Medical raises $110M for heart valve with robotic system
The startup’s third round of financing includes support from Intuitive Surgical’s venture capital arm.
By Susan Kelly • Dec. 12, 2024 -
Artivion says cybersecurity incident has disrupted order, shipping processes
The heart device manufacturer has taken certain systems offline in response to the incident, which involved the acquisition and encryption of files.
By Nick Paul Taylor • Dec. 11, 2024 -
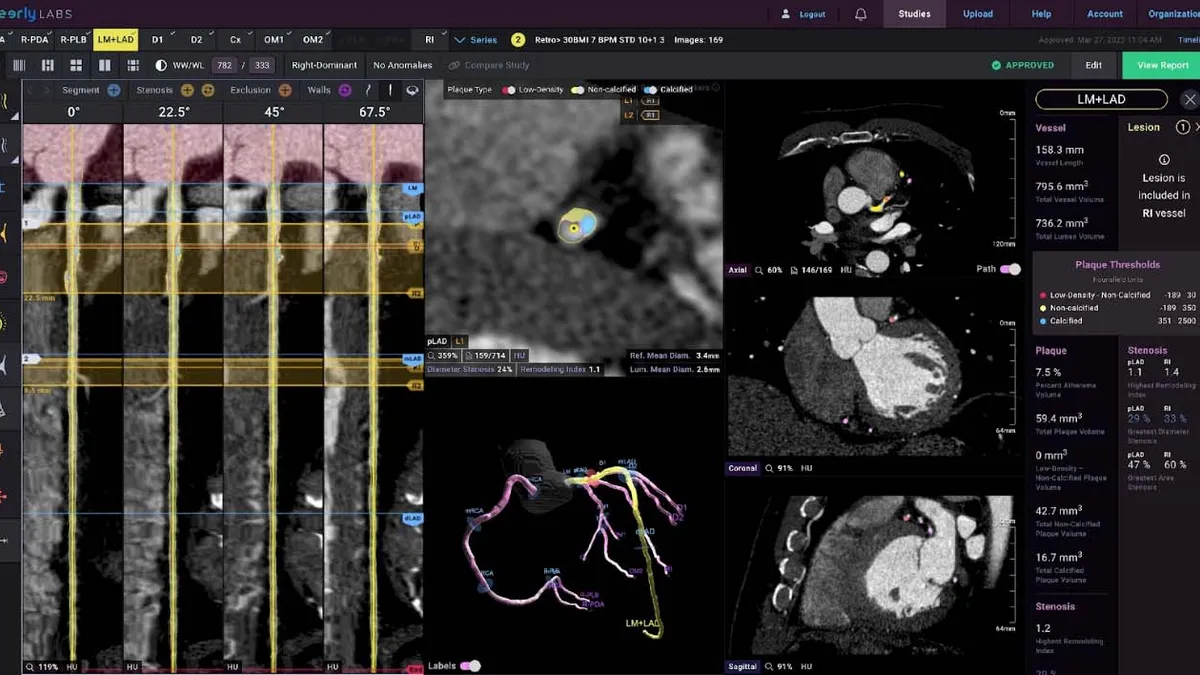
 Retrieved from Cleerly on December 06, 2024
Retrieved from Cleerly on December 06, 2024
Cleerly raises $106M to fund AI-driven heart disease tool
The company has generated evidence that shows its system is better than experts at assessing vessel narrowing, plaque volume and composition.
By Nick Paul Taylor • Dec. 6, 2024 -
Edwards expects new TAVR approval in mid-2025 to boost sales
U.S. approval of an expanded indication for transcatheter aortic valve replacement in patients without symptoms is among the growth catalysts Edwards outlined in an investor day presentation.
By Susan Kelly • Dec. 5, 2024 -
Haemonetics to offload whole-blood business for up to $67M
Selling the assets to Italy’s GVS should help Haemonetics improve its revenue growth and profit margins, analysts said.
By Susan Kelly • Dec. 4, 2024 -
Biden administration proposes Medicare, Medicaid coverage of pricey weight loss drugs
About 7.4 million Americans with obesity could have insurance coverage for drugs like Wegovy if the Trump administration allows the rule to go into effect — at the cost of $40 billion to federal and state governments.
By Rebecca Pifer • Nov. 27, 2024