Medical Devices: Page 3
-
J&J’s Ethicon recalls stapler cartridges over issue linked to 1 death
The FDA said the device can lock, leading to adverse events including surgical delay, bleeding and death.
By Nick Paul Taylor • Updated July 29, 2025 -
Olympus, Revival Healthcare partner to start GI robotics firm
Olympus and Revival Healthcare Capital agreed to invest up to $458 million combined to co-found a company that will develop a robotic platform for gastrointestinal treatment.
By Susan Kelly • July 28, 2025 -
 Explore the Trendline➔
Explore the Trendline➔
 Sara Silbiger via Getty Images
Sara Silbiger via Getty Images Trendline
TrendlineTop 5 stories from MedTech Dive
From haphazard layoffs at the Food and Drug Administration to the industry’s current IPO environment and tracking FDA-authorized AI devices, here is a collection of top stories from MedTech Dive.
By MedTech Dive staff -
Trump tariffs
US, EU agree to lower tariffs in ‘framework’ deal
Most imports from the bloc would face a 15% tariff under the pact, U.S. President Donald Trump and European Commission President Ursula von der Leyen announced Sunday.
By Philip Neuffer • July 28, 2025 -
Sponsored by Native
Value creation through innovation and design
To lead in this era, medtech companies must expand how they innovate — combining scientific rigor with human connection, intuitive design, efficient operations and strategic clarity.
July 28, 2025 -
Trump tariffs
EU preps latest round of US tariff countermeasures
The European Commission plans to install duties of up to 30%, starting Aug. 7, if the bloc cannot reach a deal with the U.S. by the end of the month.
By Philip Neuffer • July 25, 2025 -
Edwards loses longtime TAVR leader Larry Wood
Wood, who helped build Edwards’ TAVR business, will become CEO of Procept BioRobotics. Edwards also raised its sales and earnings outlook for the full year.
By Susan Kelly • July 25, 2025 -
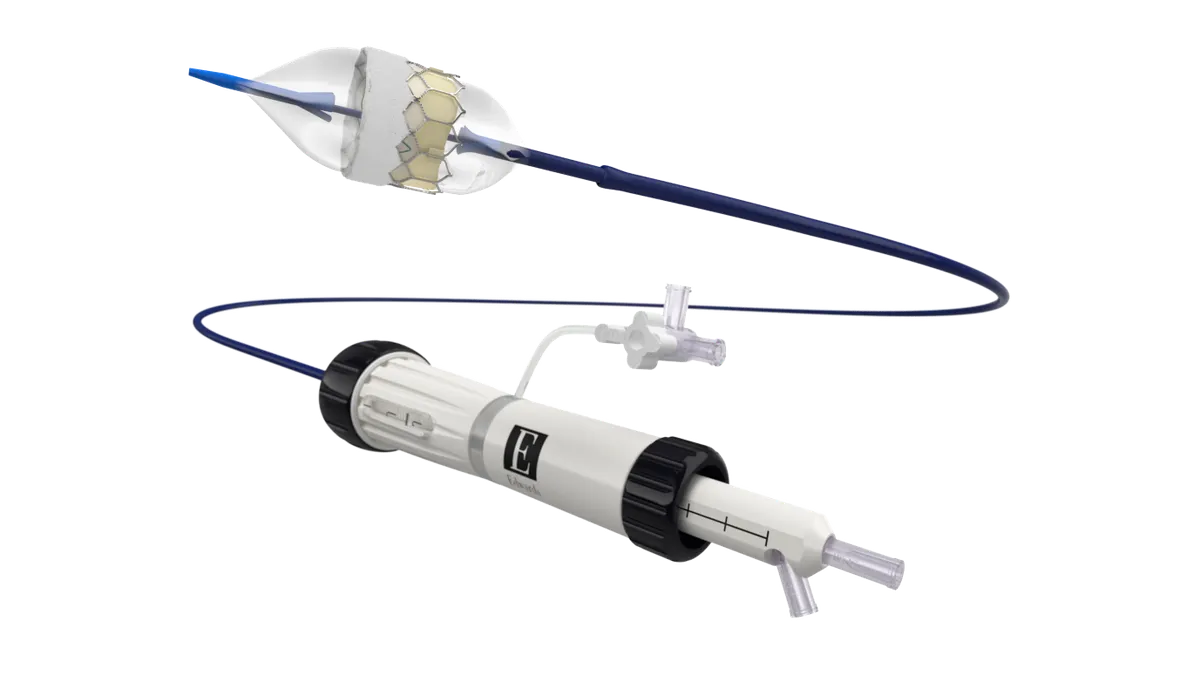
Edwards recalls arterial cannulas over exposed wires
Exposed wires could puncture the artery and cause bleeding, inadequate perfusion and hemolysis, the FDA said in its Class I recall notice.
By Nick Paul Taylor • July 25, 2025 -
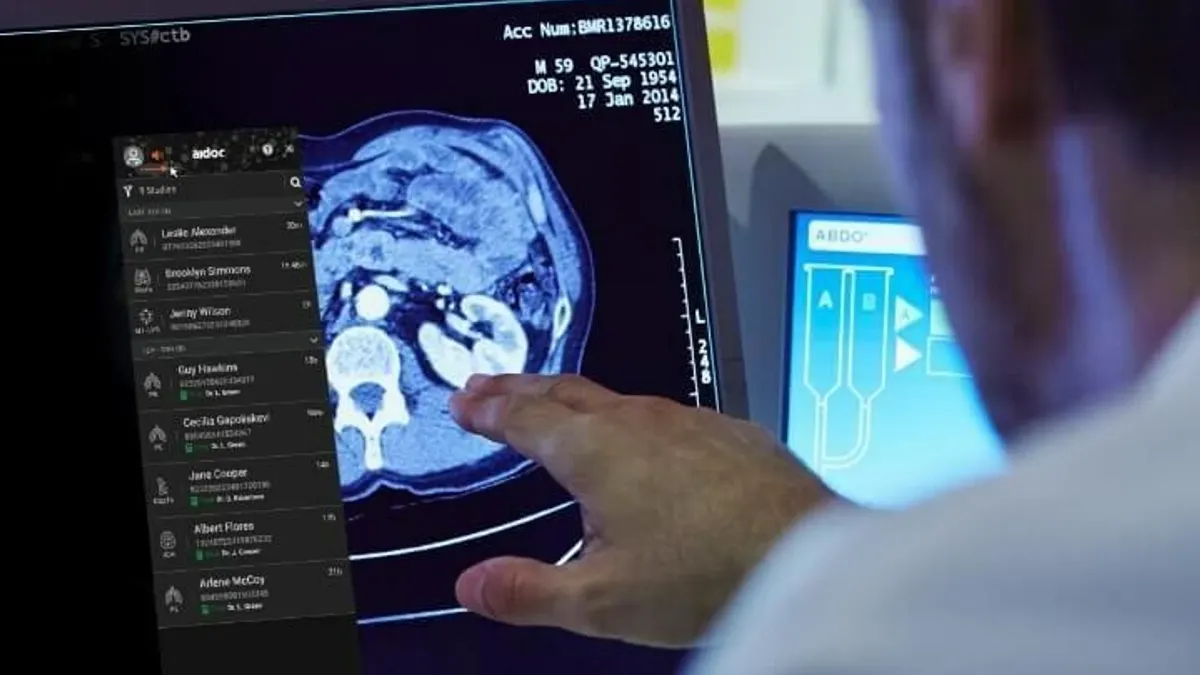
Aidoc raises $150M for AI foundation model
The technology, called CARE, is expected to allow for faster development of AI models that can cover more health conditions.
By Elise Reuter • July 24, 2025 -
Medtronic receives Type 2 label expansion for MiniMed 780G in Europe
The CE mark also allows Medtronic’s automated insulin delivery system to be used during pregnancy and by children as young as 2.
By Elise Reuter • July 23, 2025 -
Earnings & Tariffs
Intuitive Surgical tempers tariff impact expectations
Executives also addressed Medicaid concerns and competition from third-party suppliers of remanufactured surgical instruments on an earnings call.
By Susan Kelly • July 23, 2025 -
Baxter recalls certain Novum pumps over issues tied to 79 injuries, 2 deaths
The company has advised users to change the pump and infusion set at a time when a delay in treatment would not harm the patient.
By Nick Paul Taylor • July 23, 2025 -
ZimVie to sell to investment firm for about $730M
The announcement follows reports that the dental equipment company was considering a sale after receiving takeover offers.
By Elise Reuter • July 22, 2025 -
Globus promotes Keith Pfeil to CEO as former exec leaves for Dentsply
Truist Securities analysts said that “a surprise CEO change can always drive incremental uncertainty, and the announcement comes within months of reporting disappointing [first-quarter] results.”
By Nick Paul Taylor • July 22, 2025 -
Trump delays ethylene oxide emissions standards for device sterilizers
The proclamation would push back implementation of the EPA’s rule on the carcinogenic gas for two years as the Trump administration reconsiders several emissions standards.
By Elise Reuter • Updated July 21, 2025 -
Heartflow files for IPO
The company reported revenue of $37.2 million in the first quarter, up 39% year over year, but said it has faced challenges in achieving higher rates of adoption in the past.
By Nick Paul Taylor • July 21, 2025 -
Whoop’s FDA warning letter sparks debate over blood pressure as a wellness metric
Whoop pushed back on the warning letter, claiming its blood pressure feature for a wearable wristband is for “wellness” and should not be regulated as a medical device.
By Elise Reuter • July 18, 2025 -
Carlsmed seeks up to $103M IPO haul to fuel spine surgery growth
The company’s custom implants compete with devices from Medtronic, Johnson & Johnson and Globus Medical.
By Nick Paul Taylor • July 18, 2025 -
Integra recalls cranial drills over defect linked to 10 injuries
The defect has resulted in problems including a procedural delay, difficulty removing device fragments and bleeding, the FDA said.
By Nick Paul Taylor • July 17, 2025 -
Earnings & Tariffs
Abbott lowers sales forecast on diagnostics decline, US funding cuts
Medical device sales remained a bright spot for the company, however, with more than 20% growth in diabetes devices.
By Elise Reuter • July 17, 2025 -
News roundup
Intuitive demos telesurgery; SS Innovations marks 100 robot installs
The demonstration of a trans-Atlantic operation using Intuitive Surgical’s da Vinci 5 robot on a tissue model was a focus at the Society of Robotic Surgery meeting in France.
By Susan Kelly • July 16, 2025 -
GT Medical closes $53M round to commercialize brain tumor device
The money will also help the company complete enrollment in a randomized controlled trial of patients with newly diagnosed brain metastases.
By Nick Paul Taylor • July 16, 2025 -
Earnings & Tariffs
‘We are not rolling over’: J&J electrophysiology unit rebounds amid PFA rivalry
The division returned to growth in the second quarter after declining over the previous three months. Now, can the healthcare giant catch up with its rivals in pulsed field ablation?
By Ricky Zipp • July 16, 2025 -
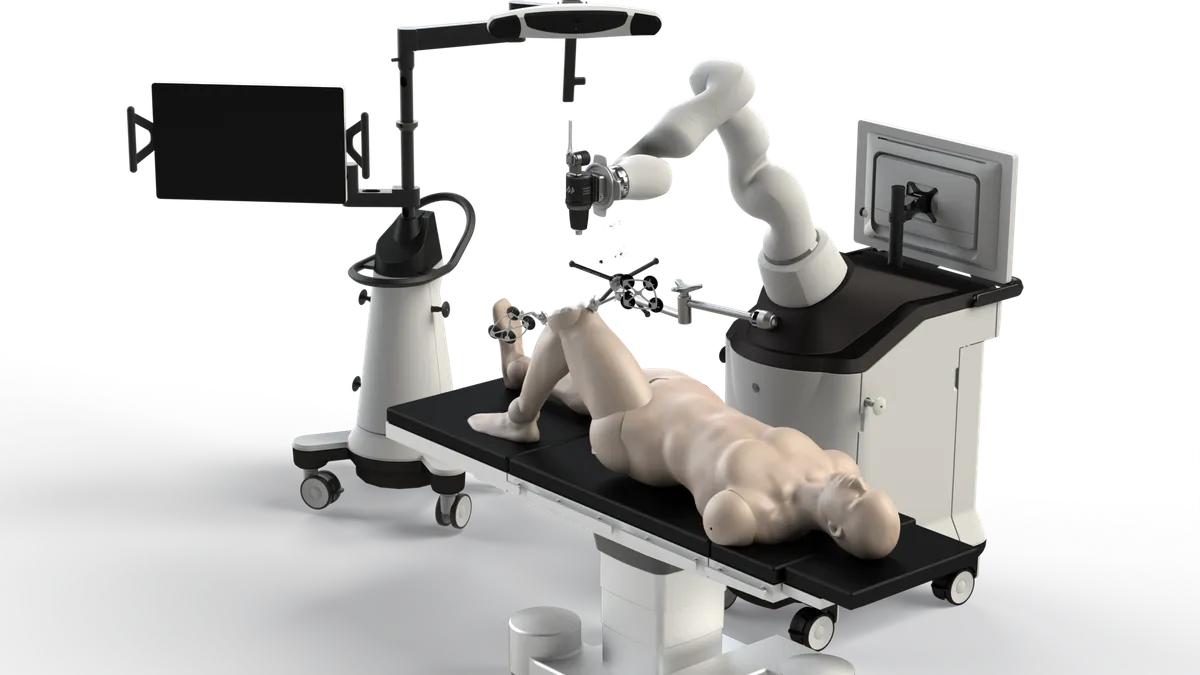
Zimmer bets on autonomous surgery with Monogram purchase
The orthopedics firm is looking to round out its robotics offerings with the planned $177 million acquisition. Some analysts questioned the value of a fully autonomous robot.
By Elise Reuter • July 16, 2025 -
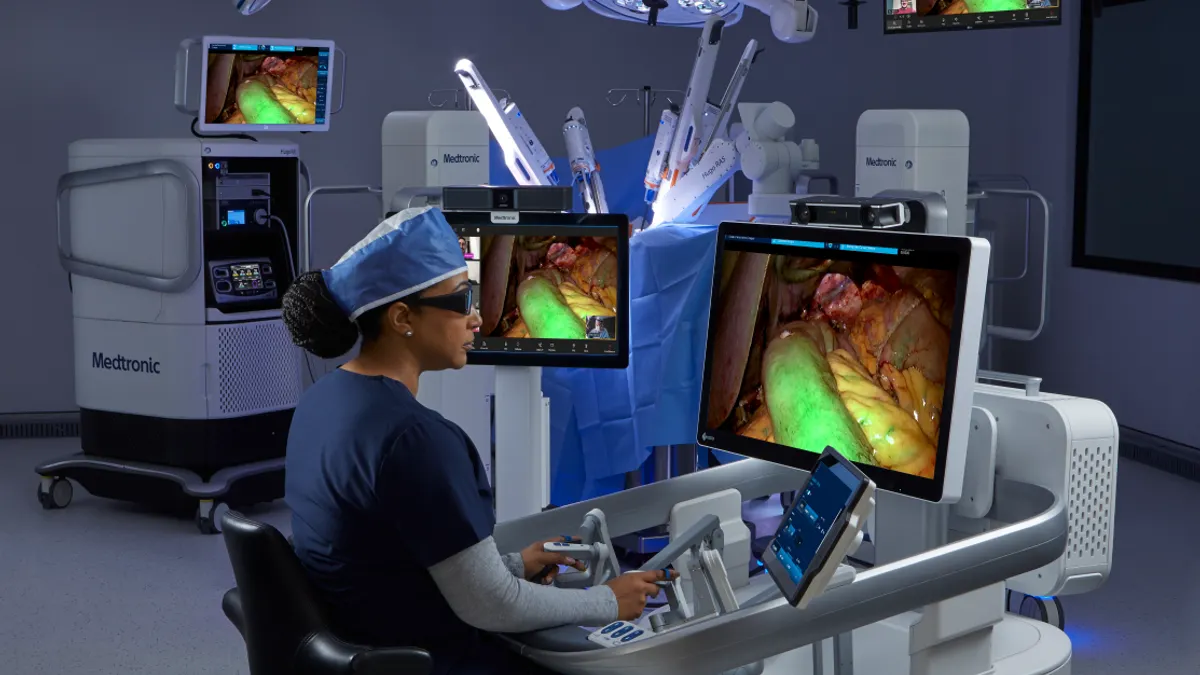
Medtronic gets CE mark for robot-driven vessel-sealing device
Medtronic said its market-leading LigaSure technology can now be used with its Hugo soft tissue robotic surgery system in Europe.
By Susan Kelly • July 15, 2025 -
Zimmer to buy robotics firm Monogram for about $177M
Monogram Technologies’ surgical robot for total knee replacements is expected to give Zimmer Biomet’s customers more options and drive robotic knee adoption in the U.S.
By Elise Reuter • July 14, 2025