Recalls: Page 5
-
Vyaire Medical recalls Airlife resuscitators over defect linked to 2 death reports
The recall covers respiratory support devices made in 2017 or earlier that can fail to provide enough ventilation.
By Nick Paul Taylor • March 22, 2024 -
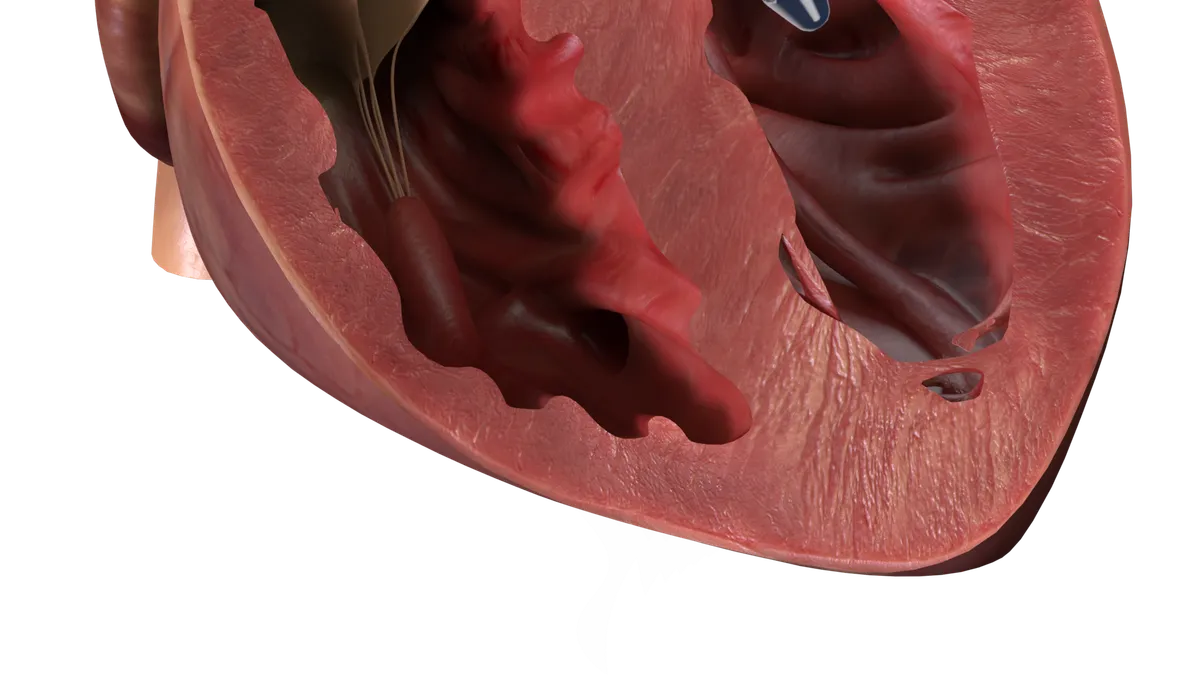
Abiomed’s latest Impella recall linked to 129 injury, 49 death reports
The J&J subsidiary warned about the risk of the catheter perforating the heart in updated instructions for use. Impella devices were part of four Class I recalls in 2023.
By Elise Reuter • March 21, 2024 -
 Explore the Trendline➔
Explore the Trendline➔
 Sarah Silbiger via Getty Images
Sarah Silbiger via Getty Images Trendline
TrendlineMedical device safety in spotlight after high profile recalls
From Philips’ massive recall of respiratory devices to ongoing health risks with breast implants, medical devices tied to patient harm have put a focus on product safety.
By MedTech Dive staff -
Deep Dive
6 ways the FDA can improve medical device recalls
Experts said improving how adverse events are tracked and requiring manufacturers to use electronic notifications could make devices safer.
By Elise Reuter • March 18, 2024 -

 Retrieved from Exactech on March 14, 2024
Retrieved from Exactech on March 14, 2024
Exactech hit with warning letter over implant packaging
The FDA found faults in the orthopedics company’s analysis of complaints demonstrating defective packaging could have accelerated wear to its implants.
By Elise Reuter • March 14, 2024 -
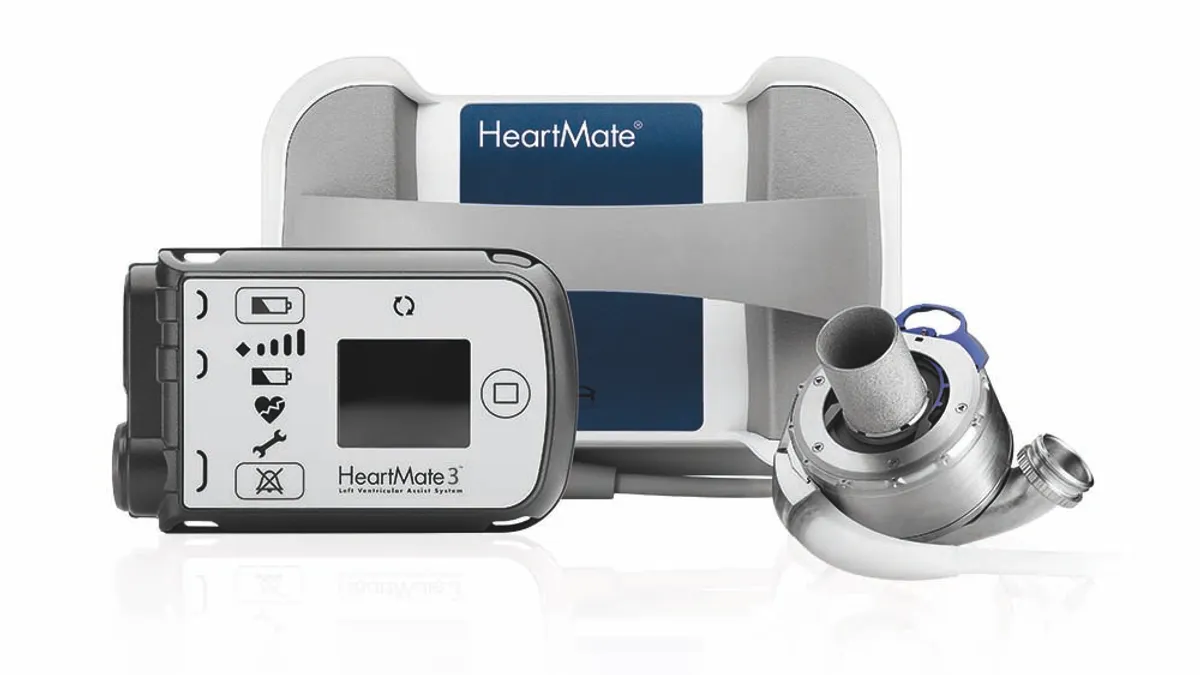
Abbott recalls Heartmate LVAD communication system
The FDA said eight reported injuries have been linked to the problem, which can cause the mechanical heart pump to unexpectedly stop or start.
By Susan Kelly • March 12, 2024 -
Medtronic recalls more than 45,000 catheter tubing units after injury reports
The issue, which is linked to 26 injuries, could result in neurological harm or death, FDA said.
By Nick Paul Taylor • March 8, 2024 -
Smiths Medical recalls nearly 86,000 syringe pumps over software problem
The company fixed the issues, which are linked to one reported injury, by updating its software.
By Nick Paul Taylor • March 6, 2024 -
Integra Lifesciences to cut about 60 jobs as it closes Indiana site
Manufacturing will be transferred to a plant in Maryland as part of a strategy to bolster operational efficiency, a company spokesperson said.
By Susan Kelly • Feb. 27, 2024 -
Medical device recall system ‘failing to meet the needs of public health,’ physicians find
Amid a GAO review of recalls, the physicians advised the watchdog to revisit unique device identifiers and clarify the FDA’s enforcement authority.
By Nick Paul Taylor • Feb. 20, 2024 -
Smiths Medical recalls syringe pumps for software malfunction
The FDA categorized the recall as Class I, noting that the software faults may cause pumps to fail, delaying or interrupting therapy.
By Nick Paul Taylor • Feb. 15, 2024 -
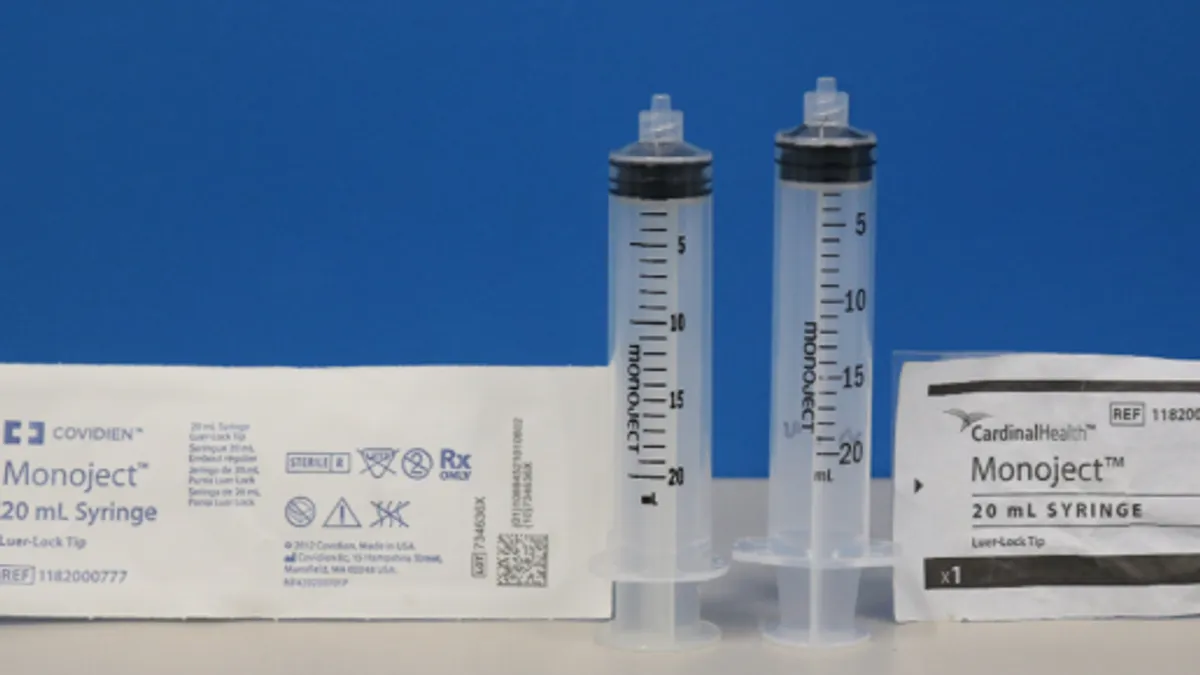
 Retrieved from Food and Drug Administration.
Retrieved from Food and Drug Administration.
Cardinal Health expands recall of Monoject syringes amid compatibility problems
The company said there have been no reports of patient deaths, but noted there is a risk of serious injury or death.
By Nick Paul Taylor • Feb. 5, 2024 -
FDA receives more reports of deaths linked to Philips’ recalled respiratory devices
From the start of July to the end of September last year, the agency received more than 7,000 reports that contained 111 deaths.
By Nick Paul Taylor • Feb. 1, 2024 -
GAO to review FDA’s oversight of medical device recalls
The government watchdog has accepted a request made in December 2023 by Sens. Richard Durbin, D-Ill., and Richard Blumenthal, D-Conn.
By Ricky Zipp • Jan. 18, 2024 -
Resmed’s magnetized masks linked to 6 patient injuries, FDA says
The company contacted customers in November after determining magnets should be kept away from active implants and metallic medical devices.
By Nick Paul Taylor • Jan. 12, 2024 -
J&J’s Megadyne restricts use of electrodes over burn risk, triggering Class I FDA notice
The J&J unit limited the use of the product in children after receiving reports of burn injuries that could be particularly harmful to them.
By Nick Paul Taylor • Jan. 9, 2024 -
Philips recalls MRI machines due to risk of explosion
The Food and Drug Administration labeled the recall as a Class I event. There has been one report of a machine exploding in the 22 years the system has been in use.
By Nick Paul Taylor • Dec. 22, 2023 -
In wake of Philips recall, senators urge review of FDA medical device oversight
Sens. Richard Durbin and Richard Blumenthal said the sleep apnea device maker “did nothing” while patients suffered.
By Susan Kelly • Dec. 14, 2023 -
Baxter sends another safety notice about syringe infusion pump errors
The company shared “reinforced guidance” to mitigate the risk of underdosing and interruptions to treatment.
By Nick Paul Taylor • Updated Dec. 6, 2023 -
BD tells customers not to use Cardinal’s Monoject syringes with Alaris pumps
The FDA labeled BD's safety notice as a Class I event. The warning comes amid several actions from the agency on the safety of syringes.
By Nick Paul Taylor • Dec. 4, 2023 -
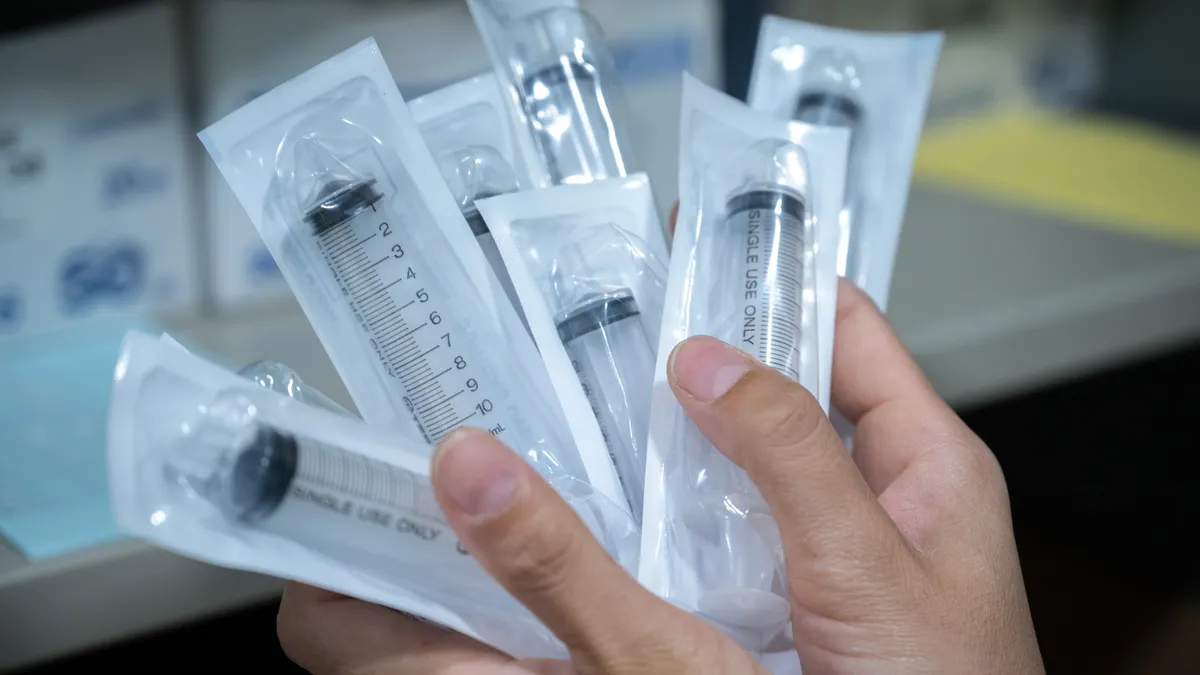
 Retrieved from Food and Drug Administration on December 01, 2023
Retrieved from Food and Drug Administration on December 01, 2023
FDA advises against using plastic syringes made in China amid reports of quality failures
Officials began the investigation after receiving information about quality issues associated with “several Chinese manufacturers of syringes.”
By Nick Paul Taylor • Dec. 1, 2023 -
Unomedical recall of infusion sets tagged as Class I by FDA
The company notified Tandem Diabetes Care, its sole consignee, in October of the risk for infusion sets to detach from insulin pumps, disrupting insulin delivery.
By Nick Paul Taylor • Nov. 28, 2023 -
FDA weighs in on SoClean’s field correction for CPAP cleaning machines
SoClean announced a voluntary field action last week to address user complaints related to improper set-up and unauthorized modifications.
By Nick Paul Taylor • Nov. 22, 2023 -
FDA concerned Cardinal Health failed to mitigate risk of incompatible syringes
Days after posting the Class 1 recall notice, the FDA said changes made to certain syringes could lead to overdose, underdose, or delays in therapy or alarms.
By Nick Paul Taylor • Nov. 21, 2023 -
B. Braun infusion pump safety correction linked to 51 complaints, one death
While no devices need to be removed, the FDA labeled the correction a Class I recall due to the risk of injury or death for patients.
By Nick Paul Taylor • Nov. 20, 2023 -

 Retrieved from Food and Drug Administration.
Retrieved from Food and Drug Administration.
Cardinal’s changes to disposable syringes trigger FDA Class I recall notice
The problem is linked to 15 reports of delayed therapy and 13 reports of inaccurate dispensing.
By Nick Paul Taylor • Nov. 15, 2023