Recalls: Page 6
-

 Retrieved from Medtronic on October 05, 2023
Retrieved from Medtronic on October 05, 2023
Stolen Medtronic laryngoscopes deemed Class 1 recall
Some of the defective devices turned up on Facebook Marketplace before the posting was taken down, the FDA said in the recall notice.
By Susan Kelly • Nov. 15, 2023 -
Olympus receives Class I recall label for another bronchoscope safety issue
The company's latest corrective action follows four reports of “endobronchial combustion” during procedures with its bronchoscopes.
By Ricky Zipp • Updated Dec. 21, 2023 -
 Explore the Trendline➔
Explore the Trendline➔
 Sarah Silbiger via Getty Images
Sarah Silbiger via Getty Images Trendline
TrendlineMedical device safety in spotlight after high profile recalls
From Philips’ massive recall of respiratory devices to ongoing health risks with breast implants, medical devices tied to patient harm have put a focus on product safety.
By MedTech Dive staff -
11 key moments in Philips’ massive recall of respiratory devices
The recall of millions of sleep apnea devices and ventilators is still ongoing more than two years later, as Philips works to repair and replace affected machines.
By Elise Reuter • Oct. 31, 2023 -
Tracker
Tracking Philips Respironics recalls
Philips recalled its Trilogy ventilators to fix new and previously reported problems with the machines. The company issued a mandatory software update and changed the devices’ instructions.
By Elise Reuter • Updated Oct. 3, 2024 -
Philips leader signed off on sale of respiratory devices despite evidence of health risks: report
Roy Jakobs approved the sale of respiratory devices held by manufacturers after the company stopped shipping new machines, according to an article published last week by Pittsburgh Post-Gazette and ProPublica.
By Nick Paul Taylor • Oct. 30, 2023 -
Fresenius’ work to replace hemodialysis tubing flagged as Class I recall by FDA
Agency officials warned healthcare professionals last year about the potential for silicone tubing to expose patients to toxic compounds.
By Nick Paul Taylor • Oct. 25, 2023 -
Olympus recall of abdominal insufflation devices linked to reports of 10 serious injuries, 1 death: FDA
The Food and Drug Administration labeled the recall a Class I event.
By Ricky Zipp • Updated Oct. 30, 2023 -
J&J’s Abiomed hit with FDA warning letter over Impella heart pump
The letter identifies quality system problems in a group of recalled devices and says monitoring software used with the pump requires premarket authorization.
By Susan Kelly • Oct. 12, 2023 -
FDA says Philips’ testing of recalled devices is inadequate
The regulator called for further tests to evaluate health risks posed to patients from the sound abatement foam used in the devices.
By Elise Reuter • Oct. 6, 2023 -
Hamilton Medical recalls ventilators for control board problem
The issue can cause a short circuit or lead the capacitor to lose function, interrupting ventilation.
By Elise Reuter • Sept. 21, 2023 -
Abbott’s neurostimulator recall, with 73 injuries reported, deemed Class I by FDA
The company contacted customers in July after receiving complaints from patients who were unable to exit the devices’ MRI mode.
By Nick Paul Taylor • Sept. 14, 2023 -
Hamilton Medical’s urgent ventilator notice deemed Class I recall by FDA
The company contacted customers in June after learning that long-term use of the devices could cause them to stop providing active ventilation.
By Nick Paul Taylor • Aug. 31, 2023 -

 Courtesy of https://news.medtronic.com/Left-Ventricular-Assist-Device-For-Advanced-Heart-Failure#assets_34137_10-122:19299
Courtesy of https://news.medtronic.com/Left-Ventricular-Assist-Device-For-Advanced-Heart-Failure#assets_34137_10-122:19299
Medtronic identifies patients at increased risk of heart pump failure
The company has recalled its HeartWare HVAD pump multiple times since it was pulled from the market in 2021.
By Elise Reuter • Aug. 29, 2023 -
Draeger’s ventilator sound insulation recall triggers FDA Class I notification
The company found concentrations of a potentially carcinogenic foam component were above acceptable levels for children.
By Nick Paul Taylor • Aug. 24, 2023 -
Integra receives warning letter related to quality issues at Boston plant
The company recalled all products made at its Boston facility over the last five years, and hopes to restart manufacturing in the fourth quarter.
By Elise Reuter • Updated Aug. 17, 2023 -
One death, 2 injuries reported in recall of Philips Trilogy ventilators
Philips warned in a previous recall about problems with the devices’ sensors that can lead to under-delivery of oxygen.
By Elise Reuter • Aug. 16, 2023 -
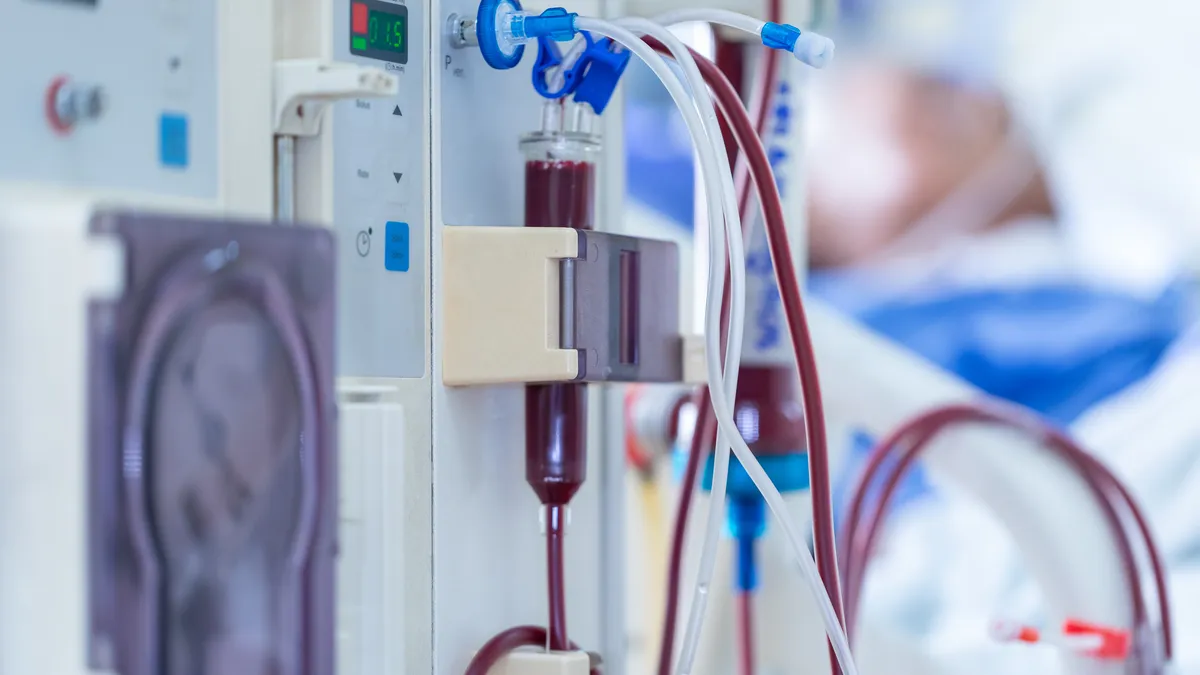
Medtronic’s dialysis device recall assigned to Class I by the FDA
The company contacted customers after routine manufacturing testing identified a blockage that could obstruct the catheter.
By Nick Paul Taylor • Aug. 16, 2023 -
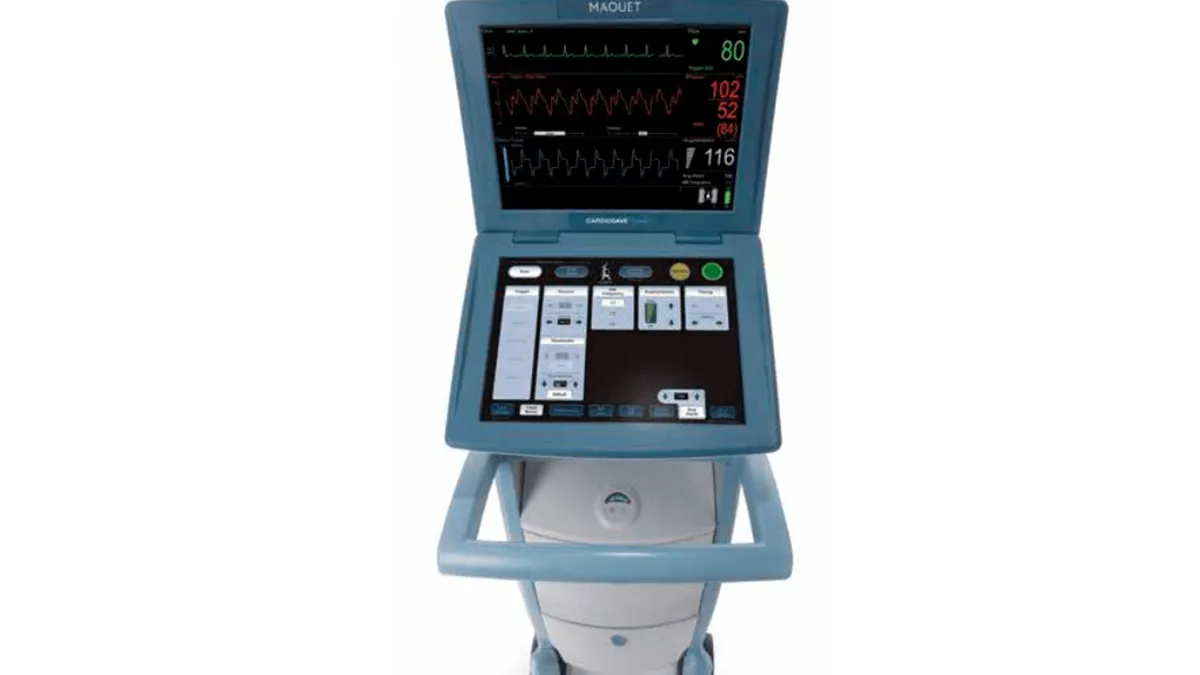
Getinge’s heart assist pump shutdown problem given FDA Class I recall tag
The company notified healthcare providers after receiving 26 complaints about Cardiosave devices, used to support severely ill patients, unexpectedly shutting down.
By Nick Paul Taylor • Aug. 11, 2023 -
Baxter recalls infusion pumps over risk of false alarms
Baxter made a software fix last year after it found pumps weren't issuing an occlusion alarm. Now, the devices might issue alarms even when there isn't a blockage.
By Elise Reuter • Aug. 2, 2023 -
Integra warns impact of plant closure will continue into 2024, with delays possible
Management has built a buffer into the plan but cautioned unforeseen events could cause its timeline to slip.
By Nick Paul Taylor • July 31, 2023 -
GE HealthCare oxygen sensor recall gets Class I label from FDA
GE warned that the sensors can limit the effectiveness of cardiac defibrillation, expose patients to electrical currents, or give inaccurate blood oxygen readings.
By Nick Paul Taylor • July 31, 2023 -
Abbott recall of heart catheter categorized as Class I event
Abbott plans to win approval for a modified steerable product and bring it to market as a replacement for the recalled device.
By Nick Paul Taylor • July 27, 2023 -
Philips reports 9% sales jump, in rebound from sleep device recall
Litigation and a U.S. Justice Department investigation into the recall are ongoing, as are discussions on a proposed consent decree.
By Susan Kelly • July 24, 2023 -
BD gains FDA clearance to relaunch Alaris infusion pump after recalls
The company will fix or replace older devices still in use at hospitals as it ramps up to resume sales in its market-leading infusion business.
By Susan Kelly • July 24, 2023 -
Four deaths attributed to interference between Abiomed’s heart pump and TAVR stents
There’s a risk that the valve implants can interact with the company’s Impella heart pumps, breaking the motor.
By Elise Reuter • Updated July 28, 2023